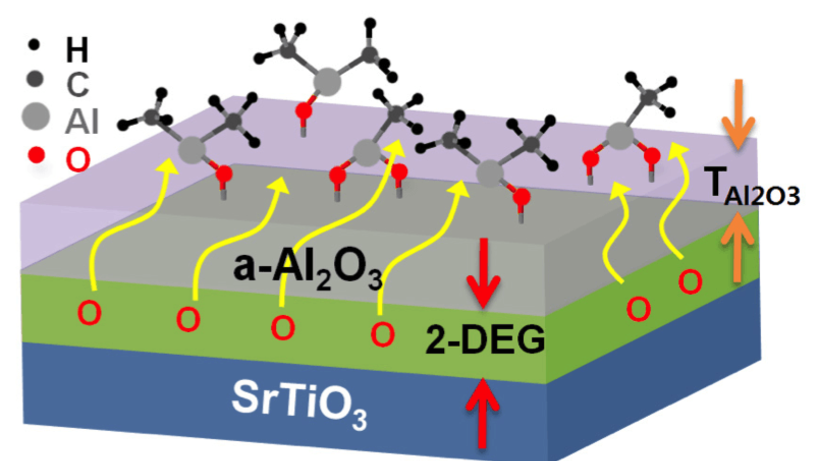
Research in our laboratory centers on thin film deposition. Typically, a compound (the "precursor") is entrained in the vapor phase into a deposition furnace, where it thermally reacts with another species (the "reactant gas") to form a thin film. Target films include thin metal films (e.g., copper, silver), thin conducting films (e.g., WN, F-SnO2), magnetic materials (e.g., Fe2O3), and high-K dielectric materials (e.g., HfO2, ZrO2).
There are three main branches to this research:
1. Precursor Design - the precursor materials are specifically designed to have high volatility and thermal reactivity at the furnace temperature. These precursors are typically inorganic or organometallic complexes containing the metal atom component of the target film.
For example, see:
Atomic Layer Deposition of Hafnium and Zirconium Oxides Using Metal Amide Precursors
Dennis M. Hausmann, Esther Kim, Jill Becker, and Roy G. Gordon
Chemistry of Materials (2002) 14: 4350-4358
2. Atomic Layer Deposition - our major focus for thin film depositions is on atomic layer deposition, or ALD. It is a process for depositing thin layers from two or more vapor precursors. The surface onto which film is to be deposited is exposed to a dose of vapor from one precursor; then any excess unreacted vapor from that precursor is pumped away. Next, a vapor dose of the reactant gas is brought to the surface and allowed to react. This cycle of steps can be repeated to build up thicker films.
For example, see:
Rapid Vapor Deposition of Highly Conformal Silica Nanolaminates
Dennis Hausmann, Jill Becker, Shenglong Wang, and Roy G.Gordon
Science (2002) 298: 402-406
3. Theoretical modeling - the deposition of smooth, even films can be difficult on complex surfaces. In particular, when deposition needs to occur in a very deep hole, it can be difficult to have the film evenly coated to the bottom. Our group has determined a theoretical model to describe the depth to which a film can be deposited in a hole, as well as presenting experimental data to verify the model.
For example, see:
A Kinetic Model for Step Coverage by Atomic Layer Deposition in Narrow Holes or Trenches
Roy G. Gordon, Dennis M. Hausmann, Esther Kim and Joseph Shepard
Chemical Vapor Deposition (2003) 9: 73-78

 ...
...